Clinical Trial Data Integrity Platform – AI-Powered eClinical System
An AI-powered clinical trial platform that eliminates manual errors, automates trial operations, and ensures complete regulatory compliance. It replaces paper-based workflows with real-time data capture, predictive monitoring, and automated submissions, helping pharma teams run faster, safer, and audit-ready trials.

Team
6–8 Members

Duration
12-16 Months

Industry
Pharmaceutical R&D / Clinical Trials / eClinical Systems / AI in Healthcare
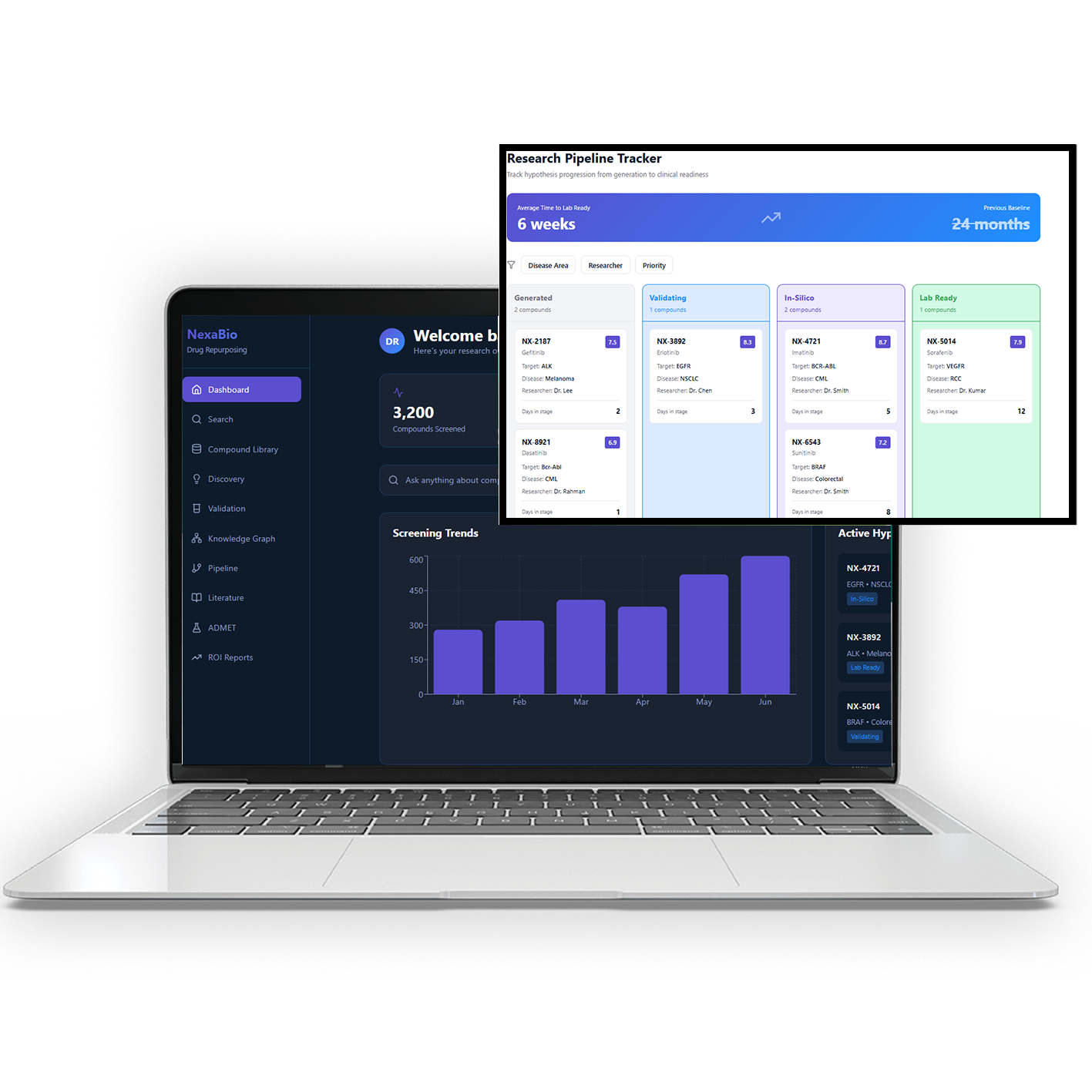

Product Overview
Clinical trials are slow and error-prone due to manual processes, fragmented data, and a lack of real-time visibility. This platform solves that by bringing all trial operations into a single system and using AI to monitor data, detect risks early, and automate workflows. It enables teams to capture data in real time, manage trials across multiple sites, and ensure compliance at every step. The result is faster trials, fewer errors, and smoother regulatory approvals.
How Does It Work?
The platform captures trial data digitally across all sites in real time, removing manual entry and reducing errors. AI continuously monitors data, identifies risks, and flags issues early, while automated workflows manage enrollment, reporting, and compliance. It also streamlines regulatory submissions, ensuring trials run faster, stay compliant, and require minimal manual effort.
User Cases
- Pharmaceutical Companies – Manage multi-site trials with better control, speed, and compliance.
- Clinical Research Organizations (CROs) – Monitor trials in real time and reduce manual workload.
- Research Sites – Capture and manage data digitally with fewer errors.
- Regulatory Teams – Ensure audit-ready data and faster submissions.
- Decentralized Trials – Enable remote participation with digital tools and real-time tracking.
Benefits
- Faster Trial Execution – Reduces overall trial timelines by 40%.
- Higher Data Accuracy – Lowers data error rates to just 0.3% with digital capture.
- Reduced Monitoring Effort – Cuts physical site visits by 70% through AI monitoring.
- Zero Compliance Issues – Achieves zero FDA data integrity findings.
- Faster Patient Enrollment – Improves recruitment speed by 45%.
- Significant Cost Savings – Saves $4.1M across active trials with 460% ROI.
- Electronic Data Capture (EDC) – FCaptures trial data in real time across all sites with built-in validation, eliminating paper-based errors.
- Digital eConsent System – Enables remote patient consent with multilingual support, secure access, and full audit tracking.
- AI Risk-Based Monitoring – Uses AI to detect risks, monitor safety, and reduce the need for physical site visits.
- Clinical Trial Management System (CTMS) – Centralizes enrollment, site performance, alerts, and trial operations.
- Regulatory Automation System – Automates eCTD submissions with FDA-compliant e-signatures and audit-ready workflows.
- AI Analytics Engine – Optimizes recruitment, predicts safety risks, and improves data quality through real-time insights.
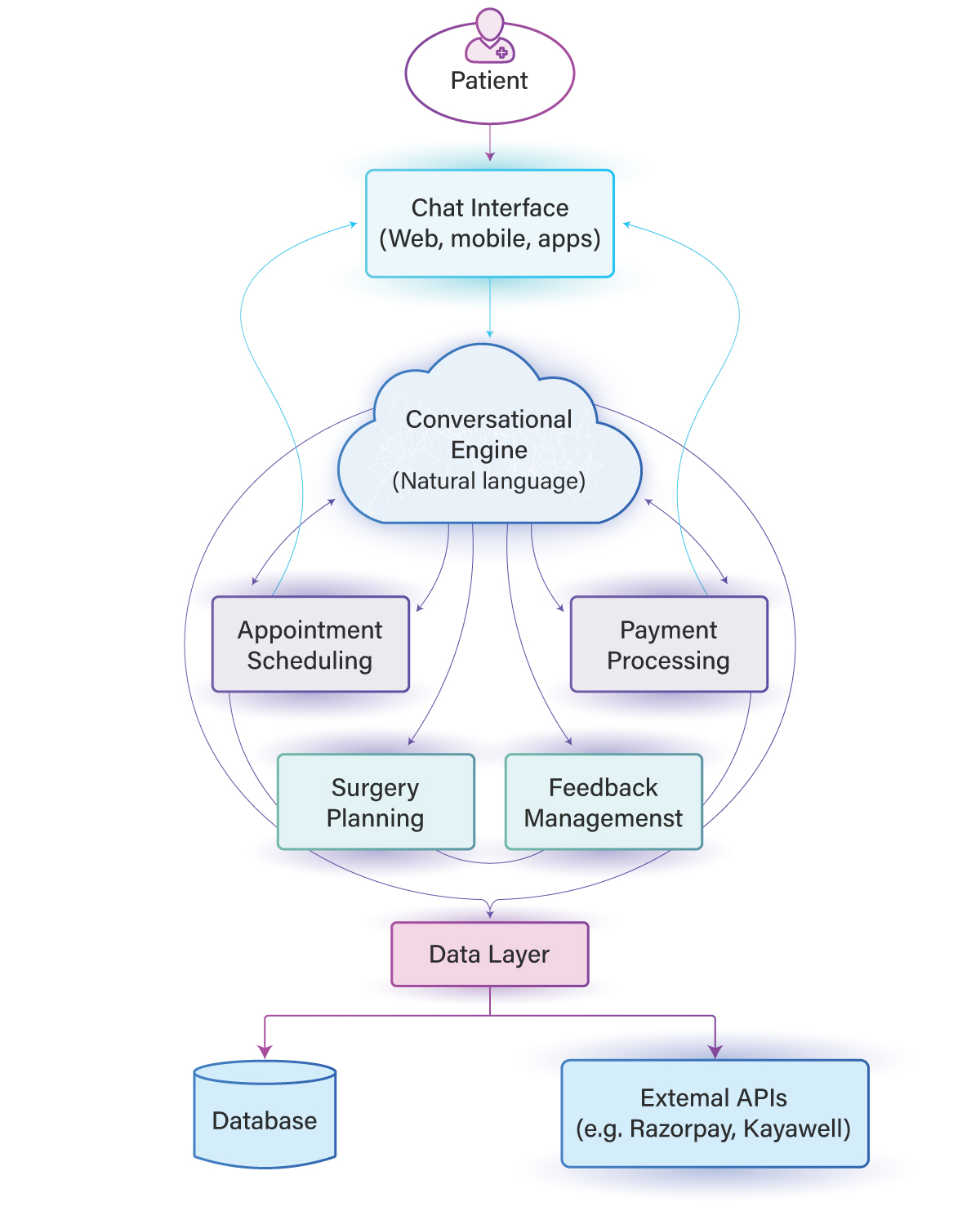
 Challenges
Challenges
Manual & Paper-Based Processes
Heavy reliance on paper CRFs and manual entry caused delays and errors.
High Data Error Rates
8–12% data errors impacted trial accuracy and compliance.
No Real-Time Monitoring
Lack of visibility delayed safety detection and decision-making.
Compliance & Audit Risks
Gaps in audit trails increase the risk of regulatory issues.
 Solutions
Solutions
End-to-End Digital Data Capture
Replaced paper with real-time EDC and validation checks to eliminate errors.
AI-Powered Monitoring
Enabled real-time risk detection, safety alerts, and reduced site visits.
Centralized Trial Management
Unified enrollment, tracking, and performance across all sites.
Built-In Regulatory Compliance
Automated submissions with full audit trails and FDA-compliant workflows.
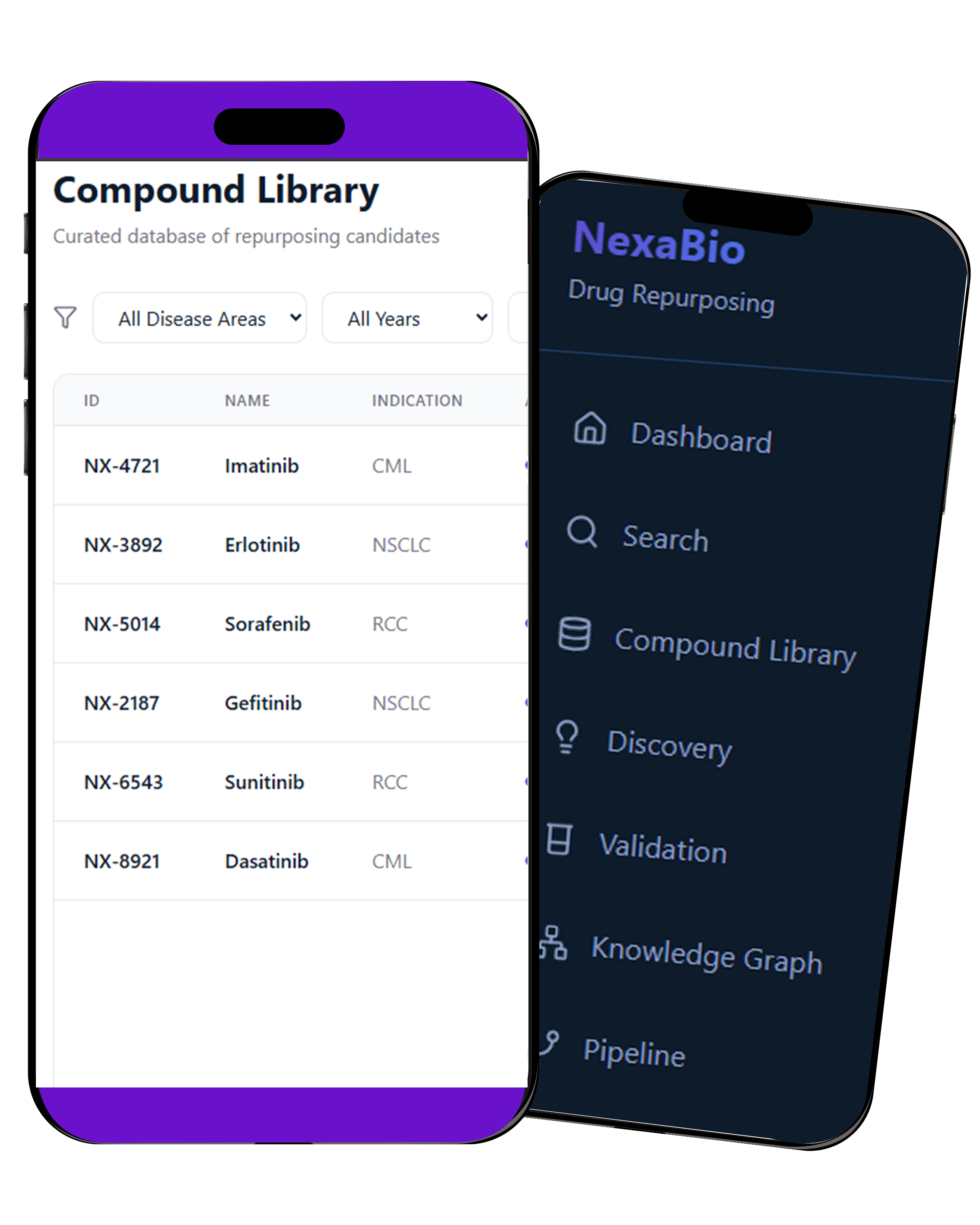
Features
Real-Time Data Capture (EDC) – Collects and validates trial data instantly across all sites.
AI Risk-Based Monitoring – Detects safety risks and data issues early with real-time alerts.
Digital eConsent System – Enables secure, remote patient consent with full audit tracking.

Centralized Trial Management (CTMS) – Manages enrollment, site performance, and trial operations in one place.
Regulatory Automation – Automates eCTD submissions with FDA-compliant e-signatures and audit trails.
Predictive Analytics – Improves recruitment, reduces deviations, and speeds up data cleaning with AI.
DreamSoft4u
delivers AI-powered eClinical solutions that eliminate data errors, ensure compliance, and accelerate clinical trials. We help pharma teams run faster, audit-ready trials with complete data integrity and zero regulatory risk.

23+
Years Experience

1000+
Satisfied Customers


24/7
Continuous Support

249+
Professional Staff
Reduce Trial Delays and Eliminate Data Errors
Discover how AI can cut timelines, reduce costs, and improve compliance.
Schedule a Free Consultation TodayWhat Our Customers Say

Nino gorgia
SmartDocs
DreamSoft4u delivered an excellent quality of work at pocket-friendly pricing. They agreed to develop an EMR-EHR-PHR solution with our frequent change requests and bug reports during the development phase. But the team stayed polite during the communication and asked for frequent feedback. Working with them was a pleasure, and their hard work is highly appreciated.

Nitin Chooda
IntouchEMr
Considering DreamSoft4u’s more than 15 years of experience in healthcare software development, I outsourced my mHealth app’s dream project. Not only did they meet my expectations, but they also handed me over a best-in-class mHealth solution competent to combat other contenders in the market. That too at a cost-effective pricing and faster time to market.

Rick Tran
EPIC EMR Integration
Great working experience with DreamSoft4u. Very responsive and very professional. Not only do they understand your needs, but they also suggest the best possible options to expand your project reach. Happy to work with them and will surely give them my next projects!!

Kem Tolliver
MRCS
I am completely satisfied with the work done by DreamSoft4u. They delivered me the same what I expected, a bespoke quality product at budget-friendly pricing. Great team, experienced and skilled developers, highly recommended

Michel.J. Kaldesh
Cyomed
Choosing DreamSoft4u for my custom healthcare software solution was the right choice, and working with them was a pleasure. I strongly recommend DreamSoft4u for any healthcare software solution or mobile app development need as they possess vast experience and a team of experienced developers in the healthcare industry.

Rich Blanton
Sniffle
The team of DreamSoft4u is very knowledgeable in Telemedicine app development. I hired them for my telemedicine app development project, and they delivered the same as I expected. I highly appreciate DreamSoft4u’s work and recommend them as the best telemedicine app development company.







